The field of gene-editing therapy is a rapidly evolving frontier with the potential to transform the treatment of both rare and common diseases. By modifying genetic material to silence or reactivate specific genes, these therapies offer the promise of durable, lifelong health benefits. However, transitioning from laboratory discovery to clinical practice requires navigating a complex landscape of scientific, financial, and ethical challenges.
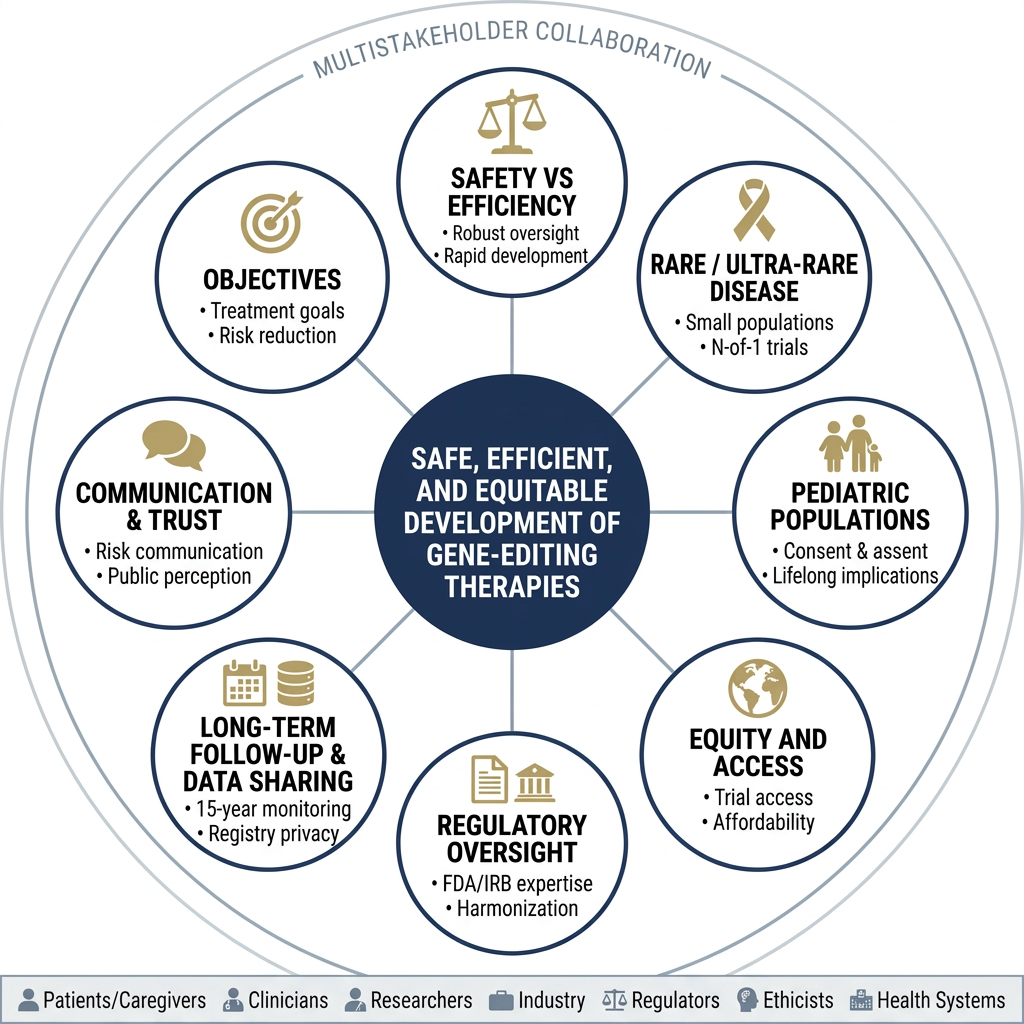
To address these hurdles, the recently established Precision Health Alliance for Genetic Medicine—a diverse consortium including patients, clinicians, industry leaders, and bioethicists—convened a specialized workshop to explore the unique ethical and regulatory concerns of gene-editing research. In a recent publication led by CVC associate faculty member Dr. Pishoy Gouda, researchers detail eight essential priorities identified during the workshop:
- Objectives: Distinguishing between life-saving cures, long-term risk reduction, and controversial human enhancements, as each requires a different threshold for acceptable safety and ethical risk.
- Competing Interests: Balancing the drive for research efficiency with rigorous safety standards to mitigate the conflict between speed and patient protection.
- Rare Diseases: Navigating “N-of-1” trials (studies focused on a single patient), which are necessary for ultra-rare conditions but challenge traditional proof-of-efficacy standards.
- Pediatric Populations: Balancing urgent early intervention with high protection standards through specialized consent models that address long-term risks.
- Equitable Access: Addressing socioeconomic barriers and funding gaps to ensure underserved populations have access to essential, life-saving therapies.
- Regulatory Oversight: Establishing international scientific standards to harmonize safety protocols and streamline global approval processes.
- Long-term Follow-up: Managing the 15-year monitoring period by addressing data privacy, registry sustainability, and the tracking of delayed adverse events.
- Communication: Building trust through transparent messaging that clarifies complex risks and the permanent nature of genetic changes.
The authors conclude that success depends on harmonizing global governance and embedding equity at every stage. Ultimately, the field must be measured not only by the conditions it cures, but by its commitment to fairness, transparency, and responsible patient protection.